|
Generally the dilution factor at each step is constant. The serial dilution method involves a stepwise dilution of a substance in solution. Basically, if diluting 1/10 and startin g off with 1 molar solution, first dilution = 0.1M, 2nd = 0.01M, 3rd = 0.001M. Therefore after a series of dilutions, you have a logarithmic curve of concentration (log10). Serial dilution is usually 1/10 dilution. This is to ensure that the exact concentration can be obtained for the experiment to become success. Serial dilution comes in handy when the solution is too concentrated to be used in experiments or ingredients preparation. There are several benefits of performing serial dilution. In serial dilution, the entire dilution factor will be multiple together to be the actual dilution factor. As example, if 1.0 ml of 3M HCl is added with 9 ml of distilled water, the dilution factor becomes 1/10. The factor can be obtained by dividing the actual volume of the initial chemical used to the final volume of the chemical after water is added. 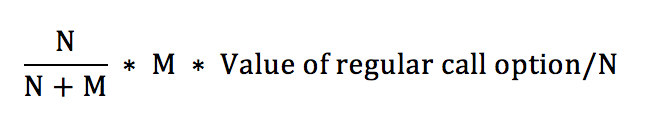
One way is by obtaining a factor called the Dilution Factor (DF).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
